“Thomas Seyfried, Ph.D.1, a professor of biology at Boston College and a leading expert and researcher in the field of cancer metabolism and nutritional ketosis, […] is one of the pioneers in the application of nutritional ketosis for cancer; a therapy that stems from the work of Dr. Otto Warburg, who was undoubtedly one of the most brilliant biochemists of the 20th century. Dr. Otto Warburg received the Nobel Prize in Physiology or Medicine in 1931 for the discovery of metabolism of malignant cells. […]
The traditionally held view or dogma is that cancer is a genetic disease, but what Warburg discovered is that cancer is really caused by a defect in the cellular energy metabolism of the cell, primarily related to the function of the mitochondria, which are the little power stations within each cell. […]
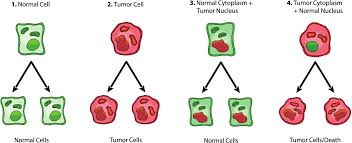
The nuclear transfer experiments basically involved transplanting the nuclei of a tumor cell into a healthy and normal cytoplasm (the material within a cell, excluding the cell nucleus), which include the mitochondria, the energy-generating organelle of the cell.
The hypothesis is that if cancer is nuclear-gene driven and the phenotype of cancer is dysregulated cell growth, meaning if genetic mutations are responsible for the observable characteristics of the disease, then those abnormal genes should be expressed in the new cytoplasm. But that’s not what happened.
Again and again, what was observed was that when the nuclei of a cancer cell were transferred into a healthy cytoplasm, the new cytoplasm did NOT form cancer. It remained healthy and normal. […]
Additional evidence has recently been produced by Benny Kaipparettu, Ph.D., and colleagues at Baylor University. When they transplanted normal mitochondria (with its nuclei intact) into cancer cell cytoplasm, it caused the cells to stop growing abnormally. It downregulated the oncogenes that were alleged to be driving the tumor and made the cells grow normally again.
On the other hand, when they took the mitochondria from a tumor cell and moved it into a very slow-growing type of cancer cell, the cancer cells began growing very rapidly. As noted by Seyfried, “When you bundle all these experiments together, you come to the conclusion that nuclear mutations cannot be the drivers of the disease.”
If genetic mutations are not the primary cause of cancer but rather a secondary, downstream effect of dysfunctional cell respiration, why and how do mutations occur? As explained by Seyfried, once the cells’ respiration is damaged, that damage then leads to a compensatory fermentation, which requires the upregulation of oncogenes (cancer genes).
Damaged respiration also produces large amounts of reactive oxygen species (ROS) and secondary free radicals that damage DNA proteins and lipids (fats inside your cellular membranes). The ROS also cause mutations in the nuclear genome. So the mutations are the result of defective respiration and subsequent exaggerated ROS production.
The ROS also target the actual mitochondria themselves, where respiration occurs, which brings us to a very important point. ROS are mostly generated through the coenzyme Q couple in the electron transport chain. Both glucose and fatty acids produce FADH2, which can generate ROS.
In contrast, fat-derived ketone bodies produce only NADH, which increases the redox span of the coenzyme Q couple and reduces production of ROS. Hence, ketone bodies are considered a more “clean” fuel than is either glucose or fatty acids. Today, most people are burning glucose as their primary fuel, thanks to an overabundance of sugar and processed grains in the diet and a deficiency in healthy fats.
If you have less ROS being generated in the mitochondria, you end up with less mitochondrial damage and less DNA damage. So not only is switching the fuel you’re feeding your body the key component of cancer treatment, but […] it’s the primary way that you prevent cancer from occurring in the first place. ”
Read the full story here, comprehensive of the related dietary approach to prevent and treat cancer
Read Dr. Seyfried’s landmark article

hello Ciliberto . I think but I’m not sure . We can therefore speak of alkaline or acidic foods ? very interesting article.
LikeLike
yes, that difference is also important. Too much acidosis contributes to disease I think.
LikeLike